Lecture Outline and Student Worksheets
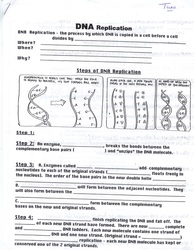
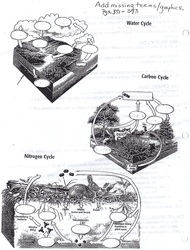
Here you can find links to some examples of worksheets used during my apprenticeship teaching. You can click on the image for an enlarged version. If you put your mouse over the picture, it will tell you how the worksheet correlated with the lesson.
| lectureoutlinehowatomsdiffer.docx | |
| File Size: | 21 kb |
| File Type: | docx |
Below is an example of a lecture outline used in Classroom Interactions as well as Student Worksheets that were assigned during my apprenticeship that I would either do with the students during lab or go over with them the next day when they completed it. There is also a Word version you can download.
The Florida Educator Accomplished Practices:
KNOWLEDGE OF SUBJECT MATTER:
8.3-Student-developed lesson plans correlated to Sunshine State Standards that address specific science content topics.
Atoms
Grade Level: 10th
How Atoms differ
SSS:
Benchmark Number: SC.912.P.8.4 Benchmark Description: Explore the scientific theory of atoms (also known as atomic theory) by describing the structure of atoms in terms of protons, neutrons and electrons, and differentiate among these particles in terms of their mass, electrical charges and locations within the atom.
Subject Area: Science Grade Level: 912 BODY OF KNOWLEDGE: Physical Science Standard: Matter -
A. A working definition of matter is that it takes up space, has mass, and has measurable properties. Matter is comprised of atomic, subatomic, and elementary particles.
B. Electrons are key to defining chemical and some physical properties, reactivity, and molecular structures. Repeating (periodic) patterns of physical and chemical properties occur among elements that define groups of elements with similar properties. The periodic table displays the repeating patterns, which are related to the atom's outermost electrons. Atoms bond with each other to form compounds.
C. In a chemical reaction, one or more reactants are transformed into one or more new products. Many factors shape the nature of products and the rates of reaction .
D. Carbon-based compounds are building-blocks of known life forms on earth and numerous useful natural and synthetic products.
HOOK:
Title: Sweet and Salty
Using Chocolate covered potato chips
What is the Key contributor in Chocolate?
Glucose
What is glucose’s molecular formula?
C6H12O6
What is the Key contributor in Potato Chips?
Salt
What is salt’s molecular formula?
NaCl
Tell the kids: These molecules have completely different tastes, even your tongue has different areas to taste sweet and salty flavors. Why is it that these things are different? Hopefully they will answer because they are made from different molecules. From here ask them what molecules are made of? Elements/atoms should be the correct answer. Then we will go into the lesson by asking them what makes atoms different.
BODY:
TEACH: Talk about the key points via powerpoint and whiteboard.
· Key ingredients in an atom: protons, neutrons, electrons.
· Atomic number = number of protons = number of electrons.
· Explain how to read different elements on the periodic table.
· Isotopes: same number of protons, but different number of neutrons.
· Mass number: sum of protons and neutrons in nucleus.
· Do some problems using atomic number and mass number to solve for number of neutrons.
o Number of neutrons = mass number – atomic number.
· Do some problems to calculate atomic mass using mass of isotopes and percent abundance
o For X1 and X2: mass contribution = (mass)(percent abundance)
o Atomic mass of X = (X1amu + X2amu)
Examples of possible questions throughout lecture: The material will be presented to the students in the powerpoint and then each sub-topic will be followed up by appropriate questions. For example: students will go over how to read the periodic table after going over definitions of protons, neutrons, electrons, and atomic number then they will be shown a picture of gold from the periodic table and asked: What and where the atomic number is? How many protons are there? How many electrons are there? The questions will be sequenced so that they will follow the sub-topic information presented to see if the students are fully grasping the concepts and where they may be struggling so the information presented can be reevaluate and presented in a way that all the students understand.
· What makes up an atom?
· What is the atomic number?
· If we are looking at hydrogen, how many protons are there?
o How many electrons?
· Looking at the periodic table, how many protons does carbon have?
o How could you tell?
· What key component makes isotopes different?
· What makes up the mass number?
· If your mass number is 22 and your atomic number is 10, how many neutrons do you have?
Review at the end of the presentation: The class will then be tested on their knowledge by working in groups and competing with one another. Questions will be put up on the powerpoint presentation and students will earn points by either finishing first and answering correctly or I will also give points when I call on groups to make sure everyone participates. Groups will be called on randomly to make sure students complete the task at hand instead of waiting to be called on to do their work. Groups will be given points and the winning group will be given a surprise! ( This will be told to the students as incentive to participate and do well. )
¨ What is the definition of atomic number?
¡ Number of protons in an atom
¨ Atomic number = ________ = _________
¡ Atomic # = # of protons=# of electrons
¨ What charge does a neutron have?
¡ None, it’s neutral
¨ If you have 12 protons, how many electrons do you have?
¡ 12
¨ An isotope has the same number of protons but different number of ___________.
¡ neutrons
¨ True or False: In nature most elements are found as a mixture of isotopes.
¡ True
¡ The unit of mass for an atom is what?
¡ Atomic mass unit (amu)
¨ Number of neutrons = ________ - _________
¡ Mass Number - Atomic Number
¨ Mass contribution from 6X = 0.451 amu
= (mass= 6.015amu)(percent abundance=7.5%)
¡ The other contributing isotope is 7X
ú Mass = 7.016
ú Abundance = 92.5% = .925
¨ What element is this??
¡ Mass contribution = (mass) (percent abundance)
¡ (7.016)(.925) = 6.490 amu
¡ SUM of mass contribution = .451 + 6.490 = 6.941 amu
¡ The element with a mass of 6.941 amu is Li
¨ The Mass Number represents the sum of what?
¡ Protons and Neutrons
¨ True or False: Protons have a negative charge.
¡ False
¨ What element has 80 protons?
¡ Mercury
¨ How many protons and electrons does Antimony have?
¡ 51
¨ An atom has 35 electrons, what is it?
¡ Bromine (Br)
¨ Potassium has a mass number of 40 and an atomic number of 19, how many neutrons does it have?
¡ Number of Neutrons = mass number – atomic number
¡ 40-19 = 21
¨ We know Silver has a mass number of 107, how many protons, neutrons, and electrons does it have?
¡ Protons and Neutrons = 47
¡ Number of Neutrons = mass number – atomic number
¡ 107-47 = 60 Neutrons
¨ True or False: The isotope Carbon-13 has 13 protons.
¡ False
¨ Atomic mass of an element is the weighted average mass of the ____________ of that element.
¡ Isotopes
¨ How many electrons does Krypton have?
¡ 36
All on Chlorine:
¨ Looking at the shortened type of notation, how many electrons does Cl have?
¡ 17
¨ What is the mass number of this isotope?
¡ 35
¨ How many neutrons does Chlorine-35 have?
¡ Number of Neutrons = mass number – atomic number
¡ 35-17 = 18
¨ How many electrons does it have?
¡ 17
A worksheet can be given out for students to attempt as homework and turned in the next day.
REMINDERS:
· Atomic number= # of protons and also # of electrons
· Isotopes vary by number of neutrons
· Mass number sum of protons and neutrons in nucleus
· Atomic mass unit =amu (almost equivalent to mass of a proton or a neutron)
· Atomic mass= weighted average of the isotopes of the element
WRAP UP:
· Make sure students can read information on the periodic table about certain elements. (Give examples and ask them random questions about elements on their periodic tables in front of them)
· Make sure calculations can be done to solve for missing pieces of information such as neutrons and atomic mass. (Have them come up to the board and do problems)
· Make sure students know vocabulary words from the lesson: atomic number, isotope, mass number, atomic mass unit (amu), and atomic mass (They will have questions presented to them continually after new sub-topics are introduced and will be tested during the review in the presentation.)
The Florida Educator Accomplished Practices:
KNOWLEDGE OF SUBJECT MATTER:
8.3-Student-developed lesson plans correlated to Sunshine State Standards that address specific science content topics.
Atoms
Grade Level: 10th
How Atoms differ
SSS:
Benchmark Number: SC.912.P.8.4 Benchmark Description: Explore the scientific theory of atoms (also known as atomic theory) by describing the structure of atoms in terms of protons, neutrons and electrons, and differentiate among these particles in terms of their mass, electrical charges and locations within the atom.
Subject Area: Science Grade Level: 912 BODY OF KNOWLEDGE: Physical Science Standard: Matter -
A. A working definition of matter is that it takes up space, has mass, and has measurable properties. Matter is comprised of atomic, subatomic, and elementary particles.
B. Electrons are key to defining chemical and some physical properties, reactivity, and molecular structures. Repeating (periodic) patterns of physical and chemical properties occur among elements that define groups of elements with similar properties. The periodic table displays the repeating patterns, which are related to the atom's outermost electrons. Atoms bond with each other to form compounds.
C. In a chemical reaction, one or more reactants are transformed into one or more new products. Many factors shape the nature of products and the rates of reaction .
D. Carbon-based compounds are building-blocks of known life forms on earth and numerous useful natural and synthetic products.
HOOK:
Title: Sweet and Salty
Using Chocolate covered potato chips
What is the Key contributor in Chocolate?
Glucose
What is glucose’s molecular formula?
C6H12O6
What is the Key contributor in Potato Chips?
Salt
What is salt’s molecular formula?
NaCl
Tell the kids: These molecules have completely different tastes, even your tongue has different areas to taste sweet and salty flavors. Why is it that these things are different? Hopefully they will answer because they are made from different molecules. From here ask them what molecules are made of? Elements/atoms should be the correct answer. Then we will go into the lesson by asking them what makes atoms different.
BODY:
TEACH: Talk about the key points via powerpoint and whiteboard.
· Key ingredients in an atom: protons, neutrons, electrons.
· Atomic number = number of protons = number of electrons.
· Explain how to read different elements on the periodic table.
· Isotopes: same number of protons, but different number of neutrons.
· Mass number: sum of protons and neutrons in nucleus.
· Do some problems using atomic number and mass number to solve for number of neutrons.
o Number of neutrons = mass number – atomic number.
· Do some problems to calculate atomic mass using mass of isotopes and percent abundance
o For X1 and X2: mass contribution = (mass)(percent abundance)
o Atomic mass of X = (X1amu + X2amu)
Examples of possible questions throughout lecture: The material will be presented to the students in the powerpoint and then each sub-topic will be followed up by appropriate questions. For example: students will go over how to read the periodic table after going over definitions of protons, neutrons, electrons, and atomic number then they will be shown a picture of gold from the periodic table and asked: What and where the atomic number is? How many protons are there? How many electrons are there? The questions will be sequenced so that they will follow the sub-topic information presented to see if the students are fully grasping the concepts and where they may be struggling so the information presented can be reevaluate and presented in a way that all the students understand.
· What makes up an atom?
· What is the atomic number?
· If we are looking at hydrogen, how many protons are there?
o How many electrons?
· Looking at the periodic table, how many protons does carbon have?
o How could you tell?
· What key component makes isotopes different?
· What makes up the mass number?
· If your mass number is 22 and your atomic number is 10, how many neutrons do you have?
Review at the end of the presentation: The class will then be tested on their knowledge by working in groups and competing with one another. Questions will be put up on the powerpoint presentation and students will earn points by either finishing first and answering correctly or I will also give points when I call on groups to make sure everyone participates. Groups will be called on randomly to make sure students complete the task at hand instead of waiting to be called on to do their work. Groups will be given points and the winning group will be given a surprise! ( This will be told to the students as incentive to participate and do well. )
¨ What is the definition of atomic number?
¡ Number of protons in an atom
¨ Atomic number = ________ = _________
¡ Atomic # = # of protons=# of electrons
¨ What charge does a neutron have?
¡ None, it’s neutral
¨ If you have 12 protons, how many electrons do you have?
¡ 12
¨ An isotope has the same number of protons but different number of ___________.
¡ neutrons
¨ True or False: In nature most elements are found as a mixture of isotopes.
¡ True
¡ The unit of mass for an atom is what?
¡ Atomic mass unit (amu)
¨ Number of neutrons = ________ - _________
¡ Mass Number - Atomic Number
¨ Mass contribution from 6X = 0.451 amu
= (mass= 6.015amu)(percent abundance=7.5%)
¡ The other contributing isotope is 7X
ú Mass = 7.016
ú Abundance = 92.5% = .925
¨ What element is this??
¡ Mass contribution = (mass) (percent abundance)
¡ (7.016)(.925) = 6.490 amu
¡ SUM of mass contribution = .451 + 6.490 = 6.941 amu
¡ The element with a mass of 6.941 amu is Li
¨ The Mass Number represents the sum of what?
¡ Protons and Neutrons
¨ True or False: Protons have a negative charge.
¡ False
¨ What element has 80 protons?
¡ Mercury
¨ How many protons and electrons does Antimony have?
¡ 51
¨ An atom has 35 electrons, what is it?
¡ Bromine (Br)
¨ Potassium has a mass number of 40 and an atomic number of 19, how many neutrons does it have?
¡ Number of Neutrons = mass number – atomic number
¡ 40-19 = 21
¨ We know Silver has a mass number of 107, how many protons, neutrons, and electrons does it have?
¡ Protons and Neutrons = 47
¡ Number of Neutrons = mass number – atomic number
¡ 107-47 = 60 Neutrons
¨ True or False: The isotope Carbon-13 has 13 protons.
¡ False
¨ Atomic mass of an element is the weighted average mass of the ____________ of that element.
¡ Isotopes
¨ How many electrons does Krypton have?
¡ 36
All on Chlorine:
¨ Looking at the shortened type of notation, how many electrons does Cl have?
¡ 17
¨ What is the mass number of this isotope?
¡ 35
¨ How many neutrons does Chlorine-35 have?
¡ Number of Neutrons = mass number – atomic number
¡ 35-17 = 18
¨ How many electrons does it have?
¡ 17
A worksheet can be given out for students to attempt as homework and turned in the next day.
REMINDERS:
· Atomic number= # of protons and also # of electrons
· Isotopes vary by number of neutrons
· Mass number sum of protons and neutrons in nucleus
· Atomic mass unit =amu (almost equivalent to mass of a proton or a neutron)
· Atomic mass= weighted average of the isotopes of the element
WRAP UP:
· Make sure students can read information on the periodic table about certain elements. (Give examples and ask them random questions about elements on their periodic tables in front of them)
· Make sure calculations can be done to solve for missing pieces of information such as neutrons and atomic mass. (Have them come up to the board and do problems)
· Make sure students know vocabulary words from the lesson: atomic number, isotope, mass number, atomic mass unit (amu), and atomic mass (They will have questions presented to them continually after new sub-topics are introduced and will be tested during the review in the presentation.)